What is Epithalon?
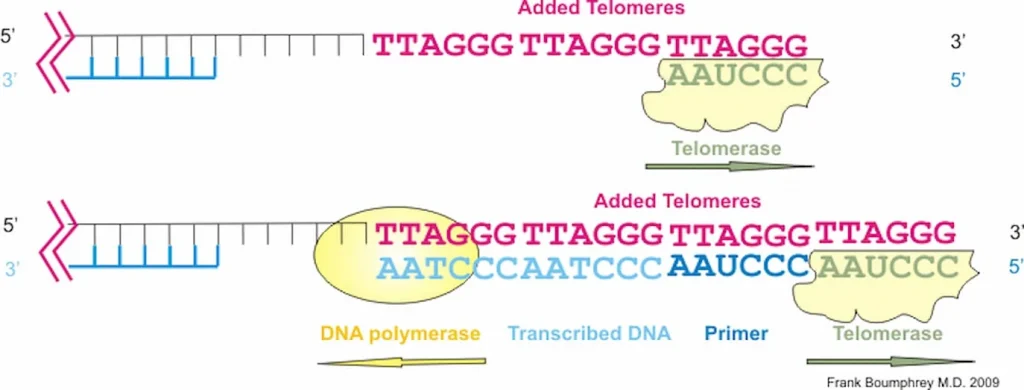
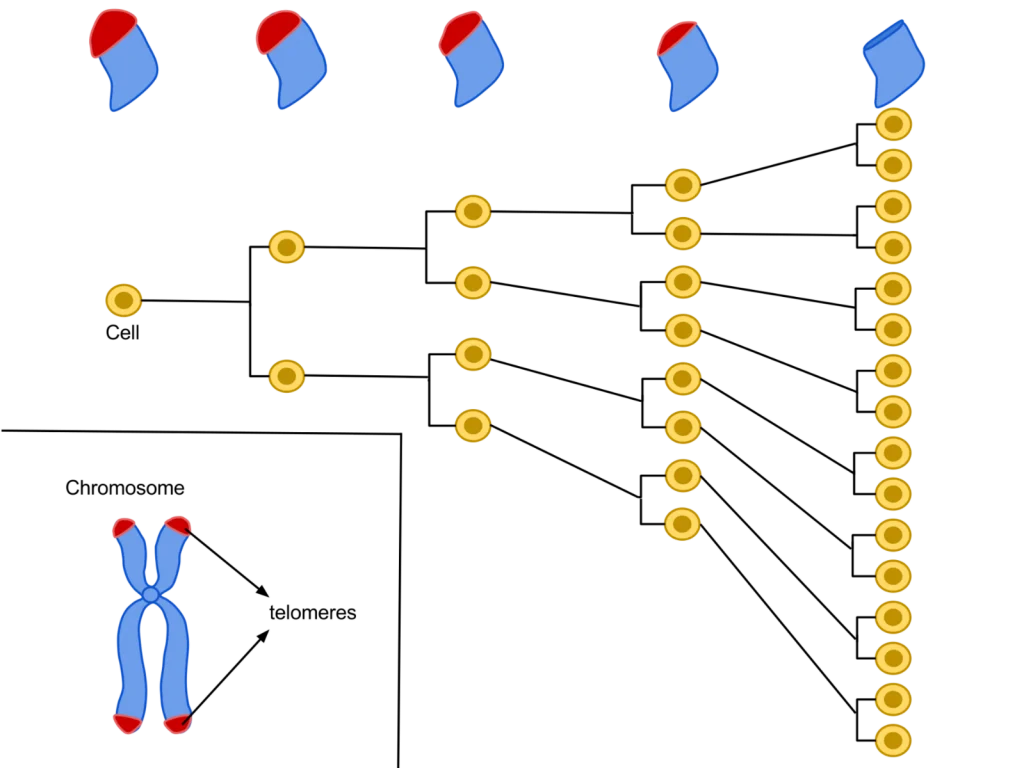
Epithalon is a short, 4 amino acid chain peptide used to regulate the cell cycle through the upregulation of telomerase activity. It has been shown to have distinctive anti-aging and anti-tumor activity across many animal and human studies. Known as the synthetic version of the tetrapeptide epithalamin, which naturally occurs in the pineal gland in our body, Epithalon (also known as Epitalon or Epithalone) was first discovered in the late 1980’s by Prof. Vladimir Khavinson from The Sankt Petersburg University, Russia.
As the most prominent tasks of the pineal gland are to maintain different kind of processes in our body, such as to normalize the activity of anterior pituitary and to maintain the levels of calcium, gonadotropins, and melatonin, its activity is highly regulated by a series of feedback mechanisms. Epithalamin acts as an antioxidant and increases the resistance to stress and lowers the levels of corticosteroids. The life extension and anti-aging properties, amongst a variety of different clinical indications, of epithalon are incredible.
Scientific research has revealed that epithalon affects the following:
– Upregulate telomerase activity
– Normalize antioxidant indices
– Reduce peroxide lipid oxidation products
– Increase activity of glutathione peroxidase
– Improve melatonin and immunity (cellular and humoral)
– Improve insulin sensitivity
– Decrease LDL and VLDL
– Improve tissue repair
– Anti-tumor effects
– Decrease mortality and increases life expectancy